UPCOMING EVENTS
View full calendarProperties of Water
LEARN ABOUT THE PROPERTIES OF WATER
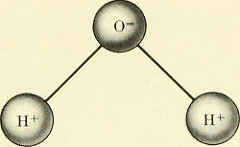
The structure of the water molecule creates weak attractions between the oxygen end of one molecule and the hydrogen ends of other water molecules. Even though these “polar” bonds are relatively weak and are constantly breaking and reforming, their existence results in many special properties of water compared to other substances on Earth. These properties of water are largely responsible for life on Earth.
Below are natural properties of water that can be significantly influenced by human activities: Chemical, Physical, and Biological.
CHEMICAL PROPERTIES
Natural waters always contain dissolved salts, micronutrients, some metals, and gases. In fact, so many substances dissolve in water that it is sometimes (mistakenly) referred to as the "Universal Solvent." While most of these substances are important for healthy aquatic ecosystems, as concentrations increase, they can have negative effects and we think of them as pollutants.
PHYSICAL PROPERTIES
BIOLOGICAL PROPERTIES
 Plants
Plants
Aquatic plants are photosynthetic organisms adapted to life in the water. Land plants are mostly “vascular” plants, having roots that take up and transport water and nutrients to leaves, where the photosynthesis occurs. Many aquatic plants have simpler structures. These “macrophytes do not need to transport water since they live in the water, and they generally can take nutrients directly from the water. The type and form that aquatic plants take depends on the water’s depth and whether the water is flowing (rivers and stream) or still (lakes, ponds or wetlands.)
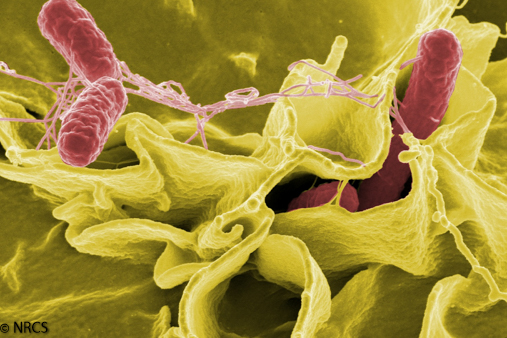 Bacteria
Bacteria
From a water quality perspective, we are mostly concerned about the role bacteria play in diseases. Bacteria and related single-celled organisms are increasingly recognized for their importance in decomposing organic material, processing minerals and nutrients, and in some cases converting carbon dioxide to new plant material.
OTHER IMPACTS


















