Artificial Carbon Sequestration in Utah
Highlights
Natural carbon storage processes cannot keep up with humans' carbon dioxide emissions.
To help rebalance Earth's carbon cycle and reduce climate warming, humans can artificially store carbon underground.
Because of their geologic features, many areas in Utah may be suitable for artificial geological carbon sequestration.
Carbon dioxide is an important greenhouse gas that helps keep Earth warm and habitable. But since humans began burning large quantities of fossil fuels during the Industrial Revolution, there has been a rapid increase in atmospheric carbon dioxide from approximately 280 parts per million in 1750 to more than 420 parts per million in 2024. Natural carbon storage processes have been unable to keep up with humans’ carbon dioxide emissions, and the sudden rise in atmospheric carbon dioxide
has led to rapid global warming and climate change.
One climate adaptation tool that may help rebalance Earth’s carbon cycle is artificial carbon storage. Geological carbon sequestration, a form of artificial carbon storage, involves capturing carbon dioxide gas and storing it within the Earth. Geological structures like deep saline aquifers, drilled oil and gas fields, subsurface sedimentary formations, subsurface folds of bedrock, buried flows of basalt from past volcanic activity, and deep coal beds can have the capacity to store carbon dioxide. Because some of these features are common in Utah, entities within the state may be well-positioned to take advantage of geological carbon sequestration.
The Carbon Dioxide Problem
Over millions of years, the natural carbon cycle has regulated the amount of carbon dioxide in the atmosphere, causing Earth’s climate to fluctuate between periods of warming, when there is more carbon dioxide in the atmosphere, and cooling, when there is less. The carbon cycle is driven by two components:
- Sources, which emit carbon. Earth’s natural carbon sources include volcanoes, fires, and animal respiration.
- Sinks, which store carbon. Natural carbon sinks include forests, soils, oceans, and chemical weathering of rocks.
Because human-caused carbon emissions have greatly outpaced Earth’s natural carbon sinks, we are currently in a period of rapid warming (Understanding Global Change, 2020). To help rebalance Earth’s carbon cycle and reduce climate warming, humans can use artificial carbon storage techniques, such as geological carbon sequestration.
How Does Geological Carbon Sequestration Work?
Geological carbon sequestration is the process of capturing carbon dioxide––either directly from the atmosphere or at the production site before it is released into the atmosphere––and then storing it within the Earth (Figure 1).
Geological Carbon Sequestration Process
Capture. Industrially produced carbon dioxide is first captured as a gas from carbon emission sources, like power-generating or cement-manufacturing facilities that burn fossil fuels (International Energy Agency, n.d.).
Compression. Once the carbon dioxide gas has been captured, it is compressed into a liquid and transported via pipes to a site that is suitable for storing the carbon dioxide underground (CLEAR Center, 2022; Szymanski, 2022; University of California Television [UCTV], 2009).
Injection. The liquid carbon dioxide is then injected at least one kilometer underground. At this depth, heat and pressure cause the carbon dioxide to undergo a phase change to a “supercritical fluid,” a fluid with characteristics of both a gas and a liquid. In this form, the carbon dioxide’s viscosity, diffusivity, and compressibility allow it to fit the area where it is being held (like a gas), yet (like a liquid) it is denser than a gas (UCTV, 2009). Because the supercritical fluid has a higher density, transforming carbon dioxide gas into this form maximizes the number of carbon dioxide molecules that can be stored (De Silva et al., 2015).

Figure 1. Movement of Carbon Dioxide During Geological Carbon Sequestration
Source: Image from the Washington Geological Survey, 2021 (Washington Department of Natural Resources)
Where Can Geological Carbon Sequestration Take Place?
Whether a site will be a good place to store carbon dioxide depends on several factors, including ground permeability, porosity, and the presence of low permeability caprocks. Permeability refers to how easily a fluid can move through a material. During geological carbon sequestration, the carbon dioxide in supercritical fluid form should be able to move throughout the rock or liquid where it is being stored. Sites with suitable porosity have holes or cracks the supercritical fluid can occupy. Sedimentary rocks like sandstone and limestone are both highly permeable and porous, making them good candidates for storing carbon dioxide (Chidsey, 2016; CLEAR Center, 2022; De Silva et al., 2015; Szymanski, 2022; UCTV, 2009).
Geological carbon sequestration also requires a caprock to limit carbon dioxide leakage during and after injection. Caprocks have low permeability and porosity. These rocks act like a “cork” by trapping carbon dioxide within the highly permeable and porous rock below. Shale is an effective caprock because it has low permeability and porosity (Chidsey, 2016; CLEAR Center, 2022; De Silva et al., 2015; Szymanski, 2022; UCTV, 2009).
In addition to permeability, porosity, and presence of a low permeability caprock, anyone intending to perform geological carbon sequestration must consider the following factors during site selection: injection techniques, pressure, temperature, storage capacity, carbon dioxide movement within the storage location, the risk of inducing small earthquakes, and the potential for harmful leakage into freshwater aquifers (Bachu, 2015). Several geological structures––deep saline aquifers, depleted oil and gas fields, subsurface sedimentary formations, large folds of bedrock, buried basalt flows, and deep coal beds––commonly meet these criteria and can support successful geological carbon sequestration (Chidsey, 2016; CLEAR Center, 2022; De Silva et al., 2015). Below, we focus on how geological carbon sequestration works in two types of sites that are relevant in Utah: deep saline aquifers and depleted oil and gas fields.
Geological Carbon Sequestration in Deep Saline Aquifers
One of the most promising locations to store carbon dioxide is in deep saline aquifers. Deep saline aquifers are underground reservoirs of extremely salty water. Because the water is so salty that it is undrinkable, using these features for carbon sequestration should have no effect on water supply (CLEAR Center, 2022; De Silva et al., 2015).
When carbon dioxide is injected into a deep saline aquifer, the resulting supercritical fluid rises to the top of the reservoir because it is less dense than the salt water. Once there, it spreads out along the bottom of the caprock, where it is constrained into small gas bubbles within the caprock’s pores. This process is known as capillary trapping (Berkeley Lab, 2010; UCTV, 2009).
Eventually, the carbon dioxide will dissolve into the water, a process known as ionic trapping. Depending on the minerals available to react within the aquifer, the dissolved carbon dioxide can precipitate out of solution, forming a solid. Ideally, the dissolved carbon dioxide precipitates out of solution as carbonate minerals, such as calcite or dolomite (Berkeley Lab, 2010; UCTV, 2009). Although forming carbonate minerals is a long and slow process, it is also the most stable form of trapping and sequestering carbon dioxide (CLEAR Center, 2022; De Silva et al., 2015; Jia et al., 2021; Szymanski, 2022). Carbonate rocks lock up carbon in terrestrial forms on million-year time scales until it is eventually released as carbon dioxide gas or dissolved carbonate ions via tectonic processes, such as uplift and weathering, metamorphism, or volcanic processes.
Geological Carbon Sequestration in Depleted Oil and Natural Gas Wells
Another place where carbon dioxide can be stored via geological carbon sequestration is in depleted oil and gas wells. Oil and natural gas are fossil fuels made of long-decayed organic matter, which, by definition, contains carbon. When decomposing organic matter is buried under Earth’s surface by natural processes, the matter––and its carbon––can get trapped in pockets. Over time, these pockets become oil and/or natural gas wells. For centuries, humans have drilled and extracted fossil fuels from these wells, proving the potential for these sites to store carbon (Chidsey, 2016; CLEAR Center, 2022). Large oil and gas fields that were developed in the 20th century but are now inactive can be suitable targets for carbon sequestration.
Geological carbon sequestration can also be combined with a technique called enhanced oil recovery that is used during fossil fuel extraction at some drilling sites. Enhanced oil recovery is when carbon dioxide is injected into active oil wells. The carbon dioxide, in its supercritical phase, loosens the oil that sticks to the permeable and porous rock and helps recover a higher percentage of oil from the well. Typically, the carbon dioxide is then allowed to leak out of the well after injection because carbon sequestration is not generally the end goal of the oil extraction practice (CLEAR Center, 2022; El-kaseeh & McCormack, 2023; Jia et al., 2021). However, some drilling operators have begun adding carbon sequestration into their enhanced oil recovery process. Combining these two practices is possible because enhanced oil recovery and carbon sequestration both require injecting carbon dioxide so it transforms into a supercritical fluid. By fusing these processes, drilling operations could improve the balance between the carbon dioxide emissions they enable and the carbon dioxide they sequester. One location that is currently applying enhanced oil recovery and geological carbon sequestration simultaneously is the Aneth oil field located in Southern Utah (Figure 2; Chidsey, 2016; Huang & Zhang, 2022).

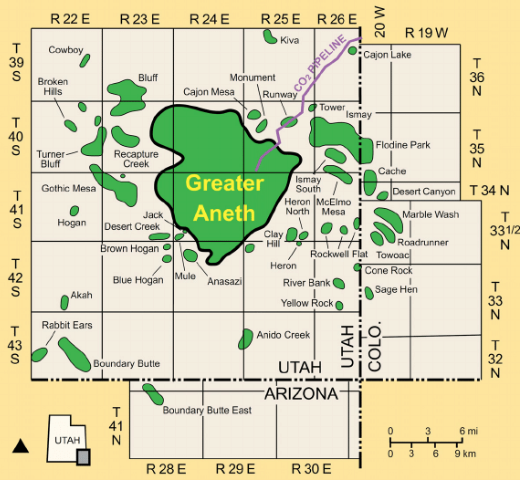
Figure 2. (left) Drilling at the Aneth Oil Field in Utah Began in 1953, and (right) Map of Greater Aneth and Carbon Dioxide Supply Pipeline
Note. Since 2005, research on simultaneous enhanced oil recovery and carbon sequestration has been possible at the Aneth oil field because it is supplied with carbon dioxide via a pipeline from a Colorado Source (Utah Geological Survery, n.d.).
Sources: (left) McPherson, 2016; (right) Slaker et al., 2013
The Aneth Research Project
Since 2005, the Utah Geological Survey and Southwest Regional Partnership for Carbon Sequestration have been conducting research on the effects of combining enhanced oil recovery and carbon sequestration at the Aneth oil field (Utah Geological Survey, n.d.). The project aims to determine whether sequestering carbon dioxide is possible while also increasing the amount of oil produced from the field. The research involves monitoring the movement of the carbon dioxide within the well and tracking the well’s ability to effectively store the carbon dioxide (Chidsey, 2016; Huang & Zhang, 2022; Szymanski, 2022; Utah Geological Survey, n.d.). By monitoring the site, risks like small earthquakes from applying pressure deep underground and carbon dioxide leakages can be understood and addressed.
Potential for Geological Carbon Sequestration in Utah
Utah has multiple geological structures, including deep saline aquifers, oil and gas reservoirs, and subsurface sedimentary formations, that could potentially sequester carbon dioxide. However, conducting geological carbon sequestration at a large scale in Utah would require installing pipelines for transporting liquid carbon dioxide to key locations around the state. Moreover, further research is needed to understand and mitigate risks associated with small earthquakes and possible water table contamination (CLEAR Center, 2022). In addition to their research on the Aneth oil field, the Utah Geological Survey is exploring other options for geological carbon sequestration throughout Utah (Szymanski, 2022). This work is important because Earth’s natural carbon storage processes can no longer keep up with the amount of carbon being emitted.
Acknowledgments
This publication was produced as part of the Climate Adaptation Intern Program (CAIP) at Utah State
University (https://qcnr.usu.edu/research/caip/). CAIP was supported by the “Secure Water Future” project that
is funded by an Agriculture and Food Research Initiative Competitive Grant (#2021-69012-35916) from the
USDA National Institute of Food and Agriculture. This fact sheet was improved based on suggestions from
Dennis Newell, Ph.D., and feedback from CAIP participants.
For correspondence, contact Scott Hotaling: scott.hotaling@usu.edu
References
- Bachu, S. (2015). Review of CO 2 storage efficiency in deep saline aquifers. International Journal of
Greenhouse Gas Control, 40, 188–202. https://doi.org/10.1016/j.ijggc.2015.01.007 - Berkeley Lab. (2010). Carbon cycle 2.0: Don DePaolo: Geo and bio sequestration [Video]. YouTube.
https://www.youtube.com/watch?v=8EqDlUMzra4 - Chidsey, T. C. (2016, January 21). Geological sequestration of carbon dioxide and enhanced oil recovery.
Utah Geological Survey. Retrieved on June 14, 2024, from https://geology.utah.gov/map-pub/survey-
notes/carbon-dioxide-sequestration/ - CLEAR Center. (2019, September 20). What is carbon sequestration and how does it work? University of
California, Davis. Retrieved on June 14, 2024, from https://clear.ucdavis.edu/explainers/what-carbon-
sequestration - De Silva, G. P. D., Ranjith, P. G., & Perera, M. S. A. (2015). Geochemical aspects of CO 2 sequestration in
deep saline aquifers: A review. Fuel, 155, 128–143. https://doi.org/10.1016/j.fuel.2015.03.0456 - El-kaseeh, G. & McCormack, K. L. (2023). Multi-scale seismic measurements for site characterization and CO 2
monitoring in an enhanced oil recovery/carbon capture, utilization, and sequestration project, Farnsworth
Field, Texas. Energies (19961073), 16(20), 7159. https://doi-org.dist.lib.usu.edu/10.3390/en16207159 - Esser, R., Levey, R., Mcpherson, B., O’Dowd, W., Litynski, J., & Plasynski, S. (2010). Preparing for a carbon
constrained world; overview of the United States regional carbon sequestration partnerships programme
and its Southwest Regional Partnership. In Geological Society, London, Petroleum Geology Conference
Series, 7(1), 1189–1195. https://doi.org/10.1144/0071189 - Huang, L. & Zhang, Z. (2022). Chapter 8: Time-lapse offset VSP monitoring at the Aneth CO 2-EOR Field. In
Geophysical Monitoring for Geologic Carbon Storage (pp. 125–144). Wiley-American Geophysical Union.
https://doi.org/10.1002/9781119156871.ch8 - International Energy Agency. (n.d.). Carbon capture, utilisation, and storage. Retrieved on June 14, 2024, from
https://www.iea.org/energy-system/carbon-capture-utilisation-and-storage - Jia, W., Xiao, T., Wu, Z., Dai, Z., & McPherson, B. (2021). Impact of mineral reactive surface area on
forecasting geological carbon sequestration in a CO 2-EOR Field. Energies, 14(6), 1608.
https://doi.org/10.3390/en14061608 - McPherson, R. S. (2016, April 26). Aneth oil field. History To Go. Retrieved on June 14, 2024, from
https://historytogo.utah.gov/aneth-oil-field/ - Milligan, M. (2019, May 8). Energy news: A partner in the CarbonSAFE Rocky Mountains Project. Utah
Geological Survey. Retrieved on June 14, 2024, from https://geology.utah.gov/map-pub/survey-
notes/energy-news/carbonsafe/ - National Energy Technology Laboratory. (n.d.). Unconventional storage. U.S. Department of Energy. Retrieved
on June 14, 2024, from https://netl.doe.gov/carbon-management/carbon-storage/unconventionalstorage - Slaker, B., Westman, E., Luxbacher, K., & Ripepi, N. (2013). Application of double-difference seismic
tomography to carbon sequestration monitoring at the Aneth Oil Field, Utah. Minerals, 3, 352–366.
10.3390/min3040352 - Szymanski, E. (2022, April 27). Assessing geologic carbon sequestration opportunities in Utah. Utah
Geological Survey. Retrieved on June 14, 2024, from https://geology.utah.gov/map-pub/survey-
notes/assessing-geologic-carbon-sequestration-opportunities-in-utah/ - Understanding Global Change. (2020, September 10). Carbon cycle. University of California Museum of
Paleontology. Retrieved on June 14, 2024, from https://ugc.berkeley.edu/background-content/carbon-cycle/ - University of California Television (UCTV). (2009, September 17). Geologic carbon sequestration [Video].
YouTube. https://www.youtube.com/watch?v=D9U0fRYrHyw - Utah Geological Survey. (n.d.). Greater Aneth Oil Field - geologic sequestration demonstration. Retrieved on
June 14, 2024, from https://geology.utah.gov/energy-minerals/ccus/aneth-oil-field/#tab-id-1 - Washington Geological Survey (2021). Geologic carbon sequestration [Fact sheet]. Washington Department of
Natural Resources. Retrieved on June 14, 2024, from https://www.dnr.wa.gov/publications/ger_fs_carbon_sequestration.pdf
July 2024
Utah State University Extension
Peer-reviewed fact sheet
Authors
Signee Storrud, Kendall Becker, Ph.D., and Scott Hotaling, Ph.D.,
Related Research