Peach Twig Borer in Utah Orchards
November 2019
Marion Murray, IPM Project Leader; Diane Alston, Entomologist (No longer at USU)

Do You Know?
- Peach twig borer is a major pest of apricot and peach/nectarine in Utah.
- Spring and early summer generations of larvae bore into and kill new shoots while later summer larvae attack fruit, typically entering fruit near the stem end.
- There are multiple generations each year.
- Use of pheromone traps and a degree-day model are used for determining accurate application timing.
- Insecticide application is the primary control; there are effective, low-toxicity options.
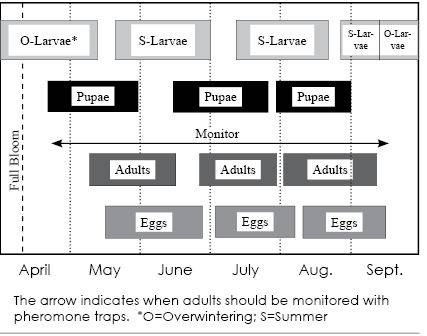
Peach twig borer (Anarsia lineatella) is found worldwide wherever stone fruits are grown. In Utah, it is a significant pest on peach/nectarine and apricot. There are typically three generations of peach twig borer in northern Utah (May-June, July, and August-September) and four or more in southern Utah. Young larvae that have overwintered emerge from protected shelters on limbs and twigs during bloom to petal-fall, and burrow into developing shoots. When populations are high, spring larval feeding can cause substantial damage to trees. The first adults are usually detected during April in southern Utah and May in northern Utah. Economic yield loss occurs during the summer when larvae of subsequent generations attack the fruit. Insecticides are currently the most effective control tactic. Lower toxicity options such as microbial products and insect growth regulators can provide excellent control when timed with early larval feeding and egg hatch.
HOSTS
apricot, peach/nectarine
almond, plum, prune
LIFE HISTORY
Larva-Overwintering Stage
- Where: First and second instar larvae found in bark cracks, crevices, and crotches in silken cells called hibernacula. (Hibernacula are small chimneys of the larva’s silk embedded with frass and wood chips.)
- When: Larvae emerge in spring (bloom to petal fall) and crawl to developing buds and terminals, where they feed inside a succulent shoot and then exit and move on to feed on additional shoots until they complete their development.
- Mature larvae of the overwintering generation pupate in protected sites on the trunk and branches.
Pupa
- Where: Located in bark cracks and crevices.
- When: Pupae are smooth, brown; without a cocoon.
- The pupae from overwintering larvae may take as long as 30 days to mature because of cool spring temperatures, while the pupation stage in summer generations is 7 - 11 days.
Adult (Moth)
- Size and Color: Moths are small, 0.3 – 0.5 inches long, with light and dark gray mottled wings. Scales on the front of the head cause the head to appear pointed.
- When: Adults of the overwintering generation emerge beginning in mid-May in northern Utah (April in southern Utah) (see Figure 1). Males and females use wing beat acoustic signals in addition to sex pheromones to locate each other for mating.
- Each mated female can lay 80 - 90 eggs.
EGG
- Where: Eggs are deposited singly on young, tender shoots, on the underside of leaves, and on developing fruit, beginning about the time of shuck fall.
- Color: Yellowish white to orange, oval shaped, and heavily sculptured.
- Hatch occurs in 4 - 18 days depending on temperature.
Larva-Damaging Stage
- Where: First generation larvae feed in the terminal shoots while summer generations attack the fruit.
- Color and Size: Young larvae are pale with light brown rings and black heads. Older larvae have a dark brown head and prothorax (segment just behind the head) and chocolate brown body. The area between body segments is lighter in color giving larvae a distinctive striped appearance. Mature larvae are about 0.5 inch long. Young larvae are pale with light brown rings and black heads. Older larvae have a dark brown head and prothorax (segment just behind the head) and chocolate brown body. The area between body segments is lighter in color giving larvae a distinctive striped appearance. Mature larvae are about 0.5 inch long.
- Larvae mature and pupate in 2-3 weeks.
- When summer generation eggs hatch, these larvae generally enter fruit to complete their development, then emerge, pupate, and become the next generation of adults.



HOST INJURY
Peach twig borer larvae are primarily attracted to new shoot growth and secondarily to maturing fruits. Larvae of early generations feed inside terminal shoots, causing the leaves to wilt and eventually kill the terminals. These dead shoots are referred to as “shoot strikes”. On young trees, repeated death of terminal branches causes stunted growth and reduced tree vigor.
As fruit pits harden (and fruit develops color), the fruits become more appealing to larvae. Larvae enter primarily through the stem end and feed just under the skin or next to the pit, especially if the pit splits open. Larvae will also enter where fruit touches another fruit, leaves, or a branch. After larvae complete development inside fruit, small exit holes may be visible. Chewed fruit is unfit for sale and is predisposed to micro-organisms that cause rotting.
TIMING CONTROL: DEGREE DAY MODEL
Insect development is temperature-dependent, so phenological events such as moth flight and egg hatch can be predicted based on accumulated heat over time, called degree days (DD). A degree day model for specific phenological events has been developed for many orchard insects, including peach twig borer. Like codling moth, peach twig borer development occurs between the lower and upper temperature thresholds of 50º F and 88° F. The degree day model is used to more accurately time and reduce the number of insecticide applications.In order for the model to work, one must first know the date of consistent moth flight, called biofix. Biofix is a biological marking point from which an insect’s development is measured for the remainder of the season. When biofix is determined, the insect model begins at 0 degree days. Biofix is determined using pheromone traps.
Large plastic delta pheromone traps are recommended to monitor adult activity. Pheromone lures are sold separately and placed on a sticky liner inside the trap to attract and trap male moths. Lures are available in a 30-day or 60-day formulation.
Determining Degree Days
To know when to hang pheromone traps, degree days are recorded starting on March 1 in northern Utah or January 1 in southern Utah. They are calculated as the area under a sine wave curve, based on the daily maximum and minimum air temperatures. For Utah, this information can be obtained from the Utah Climate Center “Utah TRAPs” online tool, for from your county extension office. (Utah TRAPs stands for Temperature Resource and Alerts for Pests).
Trap Placement
- Assemble trap with pheromone lure. Store unused lures in a freezer. To prevent contamination, do not handle or store unsealed pheromone lures together for more than one insect species, and do not reuse a trap that contained a pheromone lure from another species. Latex gloves or forceps can be used to handle lures to prevent cross-contamination.
- Place traps in orchards by early May (early April in southern Utah) or based on degree-day accumulation (see Table 1). The first moths are expected by 400 – 450 DD.
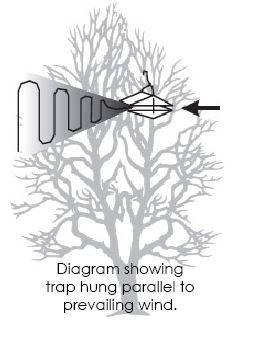
- Hang traps within the upper third of the tree canopy (preferably 6-7 ft high), making sure the trap entrance is not blocked and that it is parallel to the prevailing wind direction.
- Place one trap every 10 acres, or a minimum of two traps in each orchard.
- Hang at least one trap near the border, and one near the center of each orchard to determine if moths are immigrating from outside sources and/or overwintering within the orchard. Suspected “hot spots” within the orchard should be monitored separately.

Trap Servicing
- Check traps every 1-2 days until more than two moths are trapped on two or more consecutive nights (biofix). When determining biofix, note that zero trap captures do not necessarily mean there are no moths in the orchard. Evening temperatures below 60º F are not conducive to moth flight, and a lack of wind in the evening may prevent emission of a pheromone plume from the trap, thus no moths are lured inside.
- Once biofix has occurred, accumulated DD are reset to zero (Table 2).
- After biofix, check traps weekly. Record the number of moths caught. After recording, remove moths from the sticky liner.
- Change pheromone lures according to the label (every 30 or 60 days depending on lure type), and change sticky liners after dust, debris, or other moths have collected on the sticky surface.
- Plan to use the same type of trap and lure from year to year so that you can compare results.
- Use the recorded trap catch to evaluate the success of your control program, and to make comparisons across years.
MANAGEMENT
Cultural Controls
Prune out shoot strikes in the late spring and early summer as soon as they are detected to prevent these larvae from completing development and contributing to summer generations of the pest. If old shoot strikes are observed during pruning in late winter to early spring, plan to apply early season controls. Old strikes hold onto the dead leaves and appear as “flagged” terminals.
Biological Control
There are numerous natural enemies that will suppress peach twig borer populations. In California, several parasitic wasps (Chalcididae) and ants (Formica spp.) have destroyed a significant portion of larvae in some years.
Mating Disruption
Mating disruption (MD) is a technique that uses pheromone dispensers that permeate the air in an orchard and make it difficult for males to find females. The idea is to disrupt mating and reduce the insect population. See the USU Fact Sheet on Peach Twig Borer Mating Disruption.
This option is used by larger organic farms, especially when used in combination with organically approved insecticides such as Bacillus thuringiensis, spinosad, kaolin clay, horticultural oil, or others.
Insecticides
Treatments at the delayed dormant timing and/or during bloom are critical for reducing the severity of summer generations (Tables 2 and 3). Good coverage is important because larvae are killed after consuming insecticide residues as they tunnel in shoot twigs.
Rotate among insecticide classes (see list of insecticides in Table 4) to prevent development of resistance to insecticides in the peach twig borer population. Refer to Table 3 for recommended application timings for insecticide types.
Timing Insecticide Sprays
Studies conducted in northern Utah confirm that applications that are timed using the degree-day model (described above) can be very effective.
- The first application may be applied during the delayed dormant period (when buds swell and show the first indication of green color) using an oil plus insecticide. This treatment targets overwintering larvae.
- Research out of California has shown that the delayed dormant application plus a bloom application of a product containing Bacillus thuringiensis (Bt) is highly effective, and often no other sprays are needed. Bt has no effect on bees, but should still be applied at dawn or dusk. This treatment targets exposed larvae that are feeding on new leaves and flowers, before shoots elongate.
- If needed, the first post-bloom insecticide application should be applied from 300-360 DDs after biofix (Table 2). This timing will target 5-16% egg hatch of the first summer generation. Orchards with higher populations should use the earlier date in that range, and orchards with lower populations can use the later date.
- In some orchards, additional applications will be needed, to control the second and third summer generations. Time the cover sprays for 1200-1300 and 2140-2250 DDs after biofix, respectively (Table 2).
Table 2. Major events in a peach twig borer management program, based on accumulated degree days
| Degree Days | % Adults Emerged | % Egg Hatch | Management Event |
|---|---|---|---|
| 300 | 0 | 0 | Place traps in orchards |
| 400-450 | First moths expected | 0 | Check traps ever 1-2 days until biofix is determined |
| First Summer Generation | |||
| 0 (biofix)† |
First consistent catch | 0 | Reset degree days to 0 |
| 300-360 | 76-94 | 5-16 | Time first summer generation spray so that it is applied by this time |
| Second Summer Generation | |||
| 1200- 1300 | 46-81 | 5-16 | Time second summer generation spray so that it is applied by this time |
| Third Generation | |||
| 2140- 2250 | 37-74 | 5-16 | Time third summer generation spray so that it is applied by this time |
*Begin accumulating degree days after daily temperatures begin to exceed 50°F, typically on January 1 for southern Utah or March 1 for northern Utah.
† Biofix is when at least one moth is caught on consecutive nights.
Table 3. Cover Spray Timing and Recommendations:
| Phenological Event | Timing/ Target | Examples |
|---|---|---|
| Calyx green to first pink | Overwintering larvae | Dormant oil plus insect growth regulator (IGR) or pyrethroid* |
| Pink to Petal Fall | Overwintering larvae feeding on young shoots | Bacillus thuringiensis, spinosad, or IGR |
| Summer Sprays | Hatching larvae and adults | most options listed in Table 4* |
*Caution: Synthetic pyrethroids can cause spider mite populations to flare during hot weather by killing predaceous mites, so summer treatments should be avoided.
| MOA | Ingredient | COMMERCIAL | RESIDENTIAL | Efficacy | Notes |
|---|---|---|---|---|---|
| Conventional (non-organic) Options | |||||
| 1 | phosmet | Imidan 70 W | 3 | ||
| 1 | malathion | Malathion | Bonide Malathion; Spectracide Malathion | 2-3 | Provides just 5-7 days of protection |
| 3 | esfenvalerate | Asana XLR | Monterey Bug Buster II | 3 | |
| 3 | fenpropathrin | Danitol 2.4 ECR | 2-3 | ||
| 3 | gamma-cyhalothrin | Spectracide Triazicide | 3-4 | ||
| 3 | permethrin | Hi-yield Lawn, Garden, Pet, and Livestock | 3-4 | ||
| 3 | zeta-cypermethrin | GardenTech Sevin | 3-4 | ||
| 5 | spinetoram | Delegate WC | 3 | ||
| 5 | spinosad | Success | Bonide Captain Jack's Deadbug Brew | 3 | |
| 18 | methoxyfenozide | Intrepid 2F | 2-3 | Not registered for apricot | |
| 28 | chlorantraniliprole | Altacor | 4 | ||
| 28 | cyantraniliprole | Exirel | 4 | ||
| 3/28 | lambda-cyhalothrin/ chlorantraniliprole | Voliam XpressR | 3-4 | ||
| 4/28 | thiamethoxam/ chlorantraniliprole | Voliam Flexi | 3-4 | ||
| Organic Options | |||||
| 3 | pyrethrin | Azera, PyGanic, Tersus | Fertilome Fruit Tree Spray; Bonide Neem OiI; Monterey Take Down Garden Spray |
2-3 | Provides just 5-7 days of protection |
| 5 | spinosad | Entrust | Natural Garden Spinosad; Monterey Garden Insect Spray | 3 | Only apply if Bt was not used during bloom |
| 11 | Bacillus thuringiensis | Biobit HP, Dipel DF, XenTari | Monterey Bt, Bonide Thuricide Bt | 3-4 | Good to use during bloom because safe on bees |
| NC | Burkholderia sp. strain | Venerate XC | --- | ||
| NC | Chromobacterium subtsugae | Grandevo | 2-3 | ||
| UN | azadirachtin | Aza-Direct, | 2 | ||
Examples of brands may not be all-inclusive, but are meant to provide examples of effective insecticides registered on apple and pear in Utah.
RRestricted use products require an applicator license.















































