Fertilizer Storage

Storing fertilizer properly is a simple way to reduce point source pollution that occur from fertilizer spills and container failures.
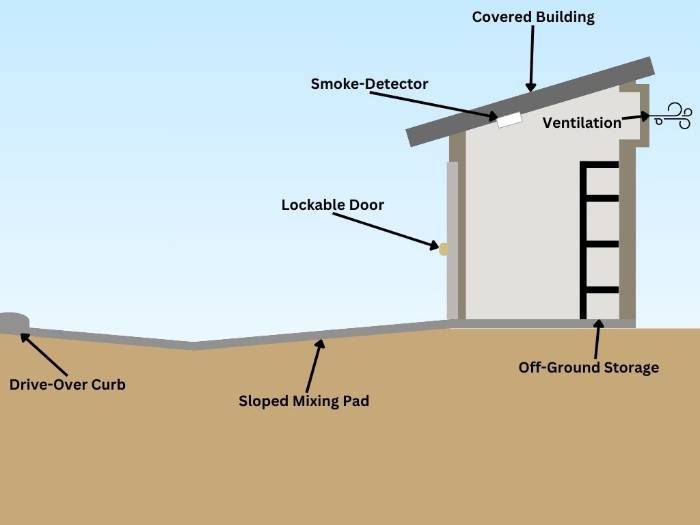
Storage Areas
A well-maintained fertilizer storage area has primary and secondary containment vessels, as well as other attributes that protect the safety of the user and prevent pollution. These areas include[3]:- The ability to be locked
- A cover to prevent stormwater runoff
- Close proximity to mixing pads
- Smoke detectors
- Ventilation
- Shelves or pallets to keep fertilizer off the ground
- An impervious surface without a drain
Fertilizer Containers
The best container to store fertilizer in is one manufactured for that purpose. Fertilizer packaging is UV resistant and the labeling contains important information regarding the application and disposal of the product[2]. Utah requires that fertilizer labels contain[5]:- The brand name
- Name and address of the manufacturer
- Directions for use
- Caution statement
- Net Weight
- A statement of guaranteed nutrients (guaranteed analysis)
- Percentage of total nitrogen, available phosphate, and soluble potash stated in the same way as in the guaranteed analysis
Mixing and Loading Pads
The location used for mixing and loading fertilizer is a critical place for preventing pollution. There is a higher potential for spills in this area. Even small quantities of fertilizer spilled frequently in the same place can cause chemicals to accumulate in the soil over time. Ideally mixing and loading should be performed on a covered impermeable surface where spilled fertilizer can be swept up or rinsed into a rinsate storage tank[3]. The surface should be large enough to park equipment and for workers to move around the equipment. The mixing or loading pad should extend at least 5 feet on each side of boom sprayers to catch splashed water[1]. A roof covering the pad lessens contamination of stormwater.
Other Considerations
Homeowners can reduce fertilizer spills, reduce waste, and keep workers safe by:- Creating an emergency readiness plan in case of a spill
- Posting important phone numbers such as the number for local county Disaster and Emergency Services office
- Training all employees and staff of procedures to reduce the environmental impact of spilled fertilizers
- Inspecting storage tanks and ensuring the container is designed for the intended use when purchasing used fertilizer tanks
- Disposing of fertilizer packaging at a sanitary landfill rather than on private property
References
[1] Hawkins, G. L., & Sumner, P. E. (2012). Pesticide storage and mixing facilities. University of Georgia Extension. https://extension.uga.edu/publications/detail.html?number=B1095&title=pesticide-storage-and-mixing-facilities[2] Larios, P. (2022, July 31). How to store fertilizers safely. Van Iperen International. https://www.vaniperen.com/how-to-store-fertilizers-safely/#:~:text=Store%20fertilizers%20in%20dry%20and
[3] Mesner, N., & Massie, L. (2012). How to protect your water from fertilizer contamination. Utah State University Extension.
[4] Trávníček, P., Kotek, L., Junga, P., Koutný, T., Novotná, J., & Vítěz, T. (2019). Prevention of accidents to storage tanks for liquid products used in agriculture. Process Safety and Environmental Protection, 128, 193–202. Science Direct. https://doi.org/10.1016/j.psep.2019.05.035
[5] Utah Code § 4-13-104
Authors
Erin Rivers, Water Quality Extension Specialist; Abby Barton, Intern
Related Research




