Parasitoid Wasps of the Invasive Brown Marmorated Stink Bug in Utah
December 2022
Zach Schumm (No longer at USU) • Kate V. Richardson • Mark Cody Holthouse (No longer at USU) • Yota Mizuno • Diane G. Alston (No longer at USU) • Lori Spears (No longer at USU)
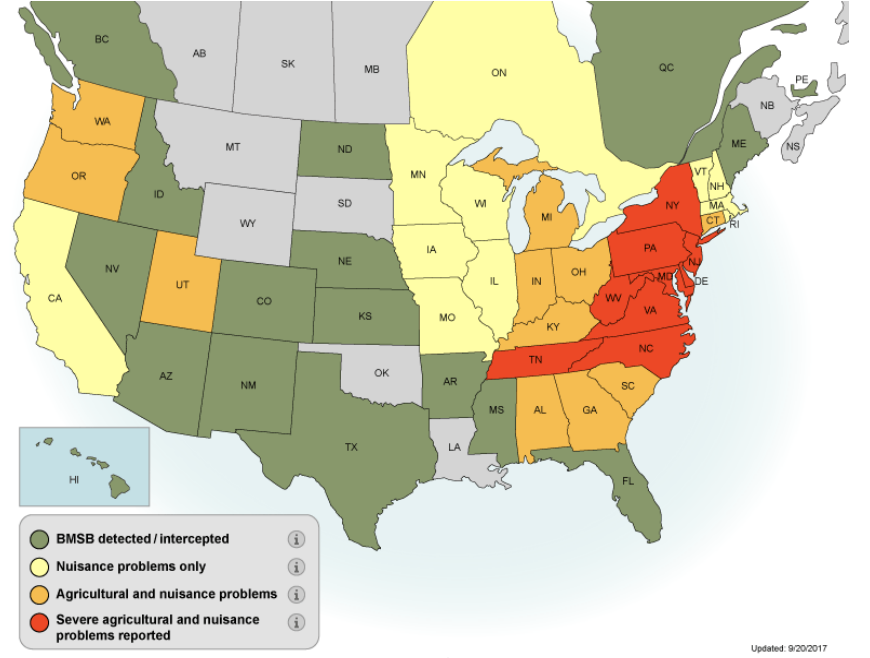
The brown marmorated stink bug (BMSB, Halyomorpha halys Stål) is an invasive agricultural and nuisance pest native to eastern Asia. It was first confirmed in the U.S. in Allentown, PA, in 1996 and has since spread to 47 U.S. states and 4 Canadian provinces, many of which have now experienced economic crop damage from this pest (Fig. 1). In Utah, BMSB is now established in six counties (Box Elder, Cache, Davis, Salt Lake, Utah, and Weber), with detection in Carbon and Kane counties.
While crop damage to peach, apple, squash, and popcorn has been observed, it is currently causing mostly nuisance problems due to overwintering adult bugs on and within human structures.
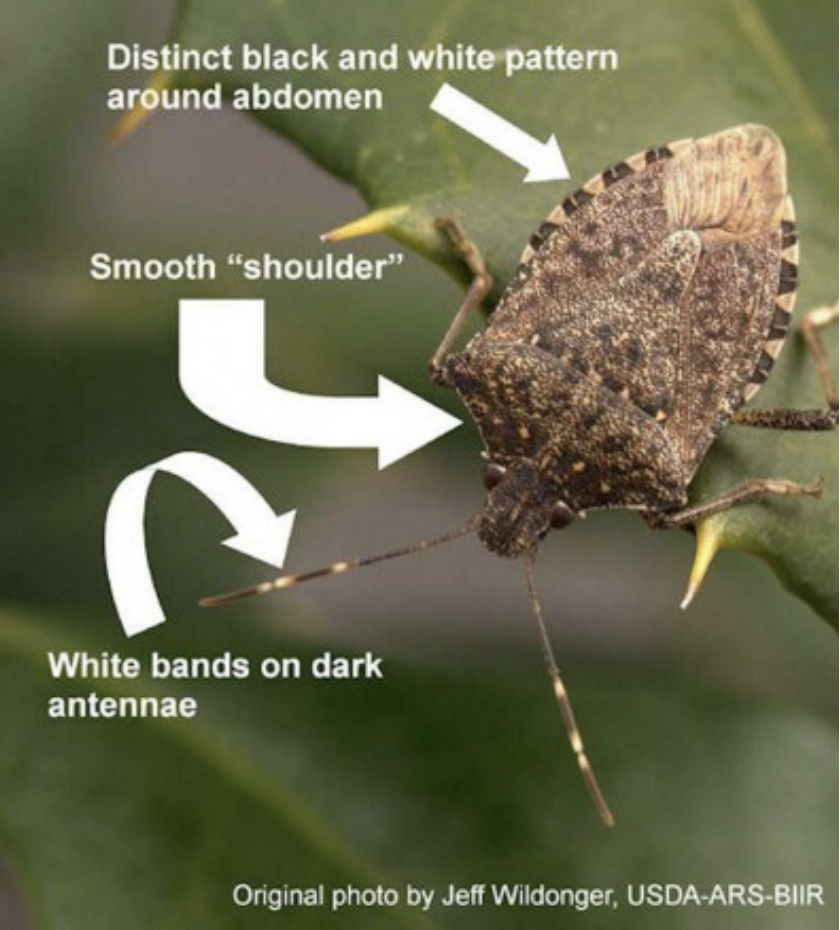
Adult BMSB are marbled brown and black, camouflaging well with woody vegetation. To separate this stink bug from native look-alikes, notice the characteristic white bands on their antennae. Native stink bug species do not have this feature. BMSB also has smooth shoulders and a black/white pattern on the edge of the abdomen (Fig. 2).
Do You Know?
- In 2012, brown marmorated stink bug (BMSB), an invasive insect pest from eastern Asia, was first detected in Utah in Salt Lake City. As of 2017, it is causing agricultural damage in northern Utah.
- There are few natural enemies of BMSB, allowing populations to increase easily.
- Parasitoid wasps that sting and kill stink bug eggs are the most promising control method. While there are native parasitoid wasps in Utah, none have been effective.
- Samurai wasp, a parasitoid native to eastern Asia, has proven to be effective at controlling BMSB populations. This wasp has been detected in some U.S. states.
BMSB is a successful invasive for a number of reasons: it is polyphagous (feeds on many plant types), highly mobile, has few natural enemies, and adults have a tough exoskeleton that is covered in a waxy, water-repellent cuticle that helps protect them from pesticide applications and the environment. Biological control, through the use of egg parasitoids, is the most suitable option for long-term management of BMSB.
General Parasitoid Information
There are at least two families of stink bug parasitoids in Utah, Eupelmidae and Scelionidae. These are small, typically black wasps that may be mistaken for small gnats or ants. They will fly in search of stink bug egg masses. Once they find the eggs, they will sting them, depositing one of their own eggs into the stink bug egg. The wasp egg will hatch, and the larva wasp will feed and develop within the stink bug egg, effectively killing the host. The adult wasp will emerge several weeks later.
The Eupelmids attacking BMSB are all generalist parasitoids, meaning that they sting the eggs of a wide variety of insects. Native parasitoids in this group are moderately successful at stinging and developing inside BMSB eggs, but are unlikely to control BMSB populations due to their generalist nature.
The second family, Scelionidae, includes some stink bug specialists, meaning they only sting stink bug eggs. Specialists are more promising as a control agent for BMSB. Although many of the native Scelionid species will sting BMSB eggs, some are unable to complete development to the adult stage. Those that can complete development within BMSB eggs have the potential to be more effective control agents due to self-propagation.
Stink bug eggs are usually bright in color (Fig. 3) and require 5 to 7 days to develop and hatch. Eggs will develop a triangular-shaped egg “burster” within shortly before stink bugs hatch from the egg (Fig. 4). However, if parasitized by a wasp, the eggs will turn dark brown or black after about a week. As the wasps develop, the eggs will continue to darken until the adult wasps emerge about 14 days later (Fig. 3).
There is usually a skewed sex ratio in emerging wasps. In a typical stink bug egg mass that consists of 14-28 eggs, one to three wasps will be male, and the rest will be female.
Male wasps will emerge first and wait for the females to emerge. Once mated, the females fly off in search of new egg masses to sting.


Parasitoid Wasp Families in Utah
Eupelmidae – Generalist Egg Parasitoids
Eupelmids are small (3-5 mm), slender wasps that are generalist egg parasitoids. One genus, Anastatus, will parasitize BMSB, as well as other stink bugs and insects. They can resemble ants at first glance. Females often have a white band or white triangles on the wings. There are three species of Anastatus known to attack BMSB in Utah (Table 1). When seen on a stink bug egg mass, a general rule is that these wasps are much larger than an individual egg (Fig. 5).
Females are typically larger than males, and under direct light can exhibit brown, green, or blue iridescence. Males are typically all black, smaller (< 4 mm), and lack wing patterns, making males indistinguishable to species without a microscope.


Scelionidae – Specialist Egg Parasitoids
Scelionids are very small (1-2 mm), but often robust wasps that are specialists on different insect groups (Fig. 7). One genus within this family, Trissolcus, only stings stink bug eggs. The wasps attacking BMSB can only be identified to species by using microscopes, as they are entirely black, small, and lack wing patterns or other characteristics to separate them with the naked eye. However, they can be generally identified in the field to family or genus using the rule that they are as small as or smaller than a stink bug egg (Fig. 8). There are at least two genera of stink bug parasitoids in the family Scelionidae in Utah (Trissolcus and Telenomus), with at least ten different species between these two genera (Table 1).


Table 1. Parasitoid wasp species found in Utah as of August 2021 from stink bug egg mass and yellow sticky card deployments. *Based on results to-date from surveys in Utah.
| Species Name | Family | Collection Method | Can Emerge from BMSB? |
|---|---|---|---|
| Anastatus mirabilis | Eupelmidae | BMSB Eggs | Yes* |
| Anastatus persalli | Eupelmidae | BMSB Eggs | Yes* |
| Anastatus reduvii | Eupelmidae | BMSB Eggs | Yes* |
| Telenomus podisi | Scelionidae | BMSB Eggs / Sticky Cards | No* |
| Trissolcus erugatus | Scelionidae | BMSB Eggs / Sticky Cards | No* |
| Trissolcus euschisti | Scelionidae | BMSB Eggs / Sticky Cards | Yes* |
| Trissolcus hullensis | Scelionidae | BMSB Eggs / Sticky Cards | Yes* |
| Trissolcus parma | Scelionidae | Sticky Cards | Unknown |
| Trissolcus thyante | Scelionidae | Sticky Cards | Unknown |
| Trissolcus strabus | Scelionidae | Sticky Cards | Unknown |
| Trissolcus utahensis | Scelionidae | BMSB Eggs / Sticky Cards | No* |
*Download pdf for actual size comparisons.
In its native range of Asia, BMSB causes minimal economic damage, presumably due to effective biological control by Trissolcus japonicus (samurai wasp) (Fig. 9). Samurai wasp was collected in China and has undergo host range-testing in U.S. quarantine facilities to assess non-target effects for release in the U.S.
However, samurai wasp has arrived on its own to the U.S. and was first detected in Delaware in 2014. It has now been found in 13 states, Canada, and many European countries as of 2021. In 2019, samurai wasp was discovered in Salt Lake City, Utah through surveys for parasitoid wasps. The samurai wasp has now been detected in six counties in Utah (Box Elder, Cache, Davis, Salt Lake, Utah, and Weber) with higher densities of wasps detected in urban landscapes.

Fig. 9. The samurai wasp (Trissolcus japonicus), a highly effective parasitoid against BMSB. It has been found in 15 U.S. states. Image courtesy of Elijah J. Talamas, USDA ARS
Surveying for Parasitoids
Methods used to survey for stink bug parasitoids include placement of stink bug egg masses on host plants in the field, finding naturally-laid stink bug egg masses laid directly on host plants, and deployment of yellow sticky cards.
Physical Egg Mass Placements:
Lab-reared stink bug eggs are attached to small squares of cardstock paper. These cards are then clipped to the underside of leaves on common hosts of stink bugs in Utah (fruit trees, vegetables, and ornamental trees such as northern catalpa [Catalpa speciosa]) (Fig. 10). Cards are deployed for 3-4 days to attract parasitoids. When collecting cards, parasitoids guarding the eggs are also collected to further assess their efficacy in stinging and developing in eggs.

Fig. 10. A BMSB egg mass clipped to a catalpa leaf with an experimental lure to attract parasitoid wasps.
Finding Naturally-laid Egg Masses:
Stink bug egg masses, laid naturally, can be found on the underside of leaves, on the fruiting structures, and occasionally on the stems of host plants (Fig. 11). Just as with deployed egg masses, parasitoids guarding the egg masses are collected to assess efficacy in killing and developing in stink bug eggs.

Fig. 11. An Anastatus wasp on naturally-laid BMSB eggs.
Yellow Sticky Cards:

Fig. 12. Yellow sticky card hung on a peach tree
to attract parasitoid wasps.
Yellow sticky cards attract various insects. Cards are deployed for two weeks on the trunks and branches of ornamental and agricultural host plants, after which the wasps are removed from the card and identified (Fig. 12). When placed in areas with high parasitoid wasp diversity, cards are an effective tool for monitoring wasp diversity and density. While cards are effective at locating parasitoid wasps, information regarding the wasp’s behavior on BMSB eggs, or their effectiveness at stinging, killing, and sustaining populations within them cannot be determined.
Enhancing Biological Control
If You See a Parasitoid Wasp:
If you see a parasitoid on an egg mass, be mindful that it is beneficial for your garden/crops, as the wasps are potentially killing the stink bugs that would normally emerge and begin eating your plants. You can choose to either leave the stink bug eggs and parasitoids on the plant (preferred), or chase off the parasitoid, remove the stink bug eggs from the plant, and freeze or destroy them before they hatch. It is possible that if the parasitoid wasps are left on the egg masses, then you may end up with more wasps, and consequently fewer stink bugs negatively impacting your plants. Parasitoids that are known to attack BMSB eggs in Utah can be seen in Figure 13.



Fig. 13. Select native parasitoid wasps stinging BMSB in Utah. From the top left: Trissolcus erugatus, Trissolcus utahensis, Anastatus reduvii, Telenomus podisi, and Trissolcus euschisti.
Reduce Pesticide Use:
Parasitoid wasps are very sensitive to insecticides, so avoid or limit the use of chemical sprays. Avoid broad-spectrum, persistent insecticides that kill many different invertebrates. Carbamates, organophosphates and pyrethroids leave harmful residues, and neonicotinoids and other systemic insecticides can poison parasitoids that feed on pollen and nectar. When needed, limit the number of chemical applications and target applications to specific problem areas.
Provide Floral Resources:
Floral resources provide a number of benefits to parasitoid wasps including attracting, feeding and sheltering them.
Flowering plants may attract parasitoids to gardens and crops. Most adult parasitoids feed on plant fluids and sugars found in the nectar and pollen of flowering plants. Access to these resources allow parasitoid wasps to survive longer and produce more progeny. The best nectar sources are often flowers with a wide or shallow corolla where wasps can easily reach the nectar. Buckwheat, sweet alyssum, basil, cilantro, and dill have specifically shown benefits to Trissolcus species.
These plants are also essential to the overall habitat of parasitoid wasps. They can provide shade, protection from predators, and overwintering sites. Consistently providing floral resources via companion planting in outdoor spaces supports parasitoid wasps and their ability to provide effective biological control.

Fig. 14. Trissolcus japonicus on buckwheat flowers.
Additional Resources
- Alston, D. G. (2011). General Concepts of Biological Control. Utah State University Extension Fact Sheet, IPM-015-11, 2. Holthouse, M. C., Alston, D. G., Spears, L. R., & Petrizzo, E. (2017). Brown marmorated stink bug [Halyomorpha halys (Stal)]. Utah State University Extension Fact Sheet, ENT-144-17, 8.
- Holthouse, M. C., Spears, L. R., & Alston, D. G. (2021a). Urban host plant utilisation by the invasive Halyomorpha halys (Stål) (Hemiptera, Pentatomidae) in northern Utah. NeoBiota, 64, 87–101.
- Holthouse, M. C., Spears, L. R., & Alston, D. G. (2021b). Comparison of yellow and blue sticky cards for detection and monitoring parasitoid wasps of the invasive Halyomorpha halys (Hemiptera: Pentatomidae). Journal of Insect Science, 21(5), 1.
- Schumm, Z. R., Alston, D. G., & Spears, L. R. (2021). Behavior of the Brown Marmorated Stink Bug, Halyomorpha halys (Stål) (Hemiptera: Pentatomidae), in the Utah Agricultural Landscape Based on Trap Captures and Visual Sampling Studies. Proceedings of the Entomological Society of Washington, 123(1), 206–216.
- Schumm, Z. R., Alston, D. G., Spears, L. R., & Manlove, K. (2020). Impact of brown marmorated stink bug (Hemiptera: Pentatomidae) feeding on tart cherry (Rosales: Rosaceae) quality and yield in Utah. Journal of Economic Entomology, 113(5), 2328–2334.
- Spears, L. R., Alston, D. G., & Murray, M. (2018). Brown marmorated stink bug management for fruits and vegetables in Utah. Utah State University Extension Fact Sheet, ENT-197-18, 6.
- Spears, L. R., Davis, R., & Ramirez, R. A. (2015). Invasive Insects Lookalikes. Utah State University Extension Fact Sheet, ENT-175-15-PR, 6.
- Talamas, E. J., Herlihy, M. V., Dieckhoff, C., Hoelmer, K. A., Buffington, M., Bon, M.-C., & Weber, D. C. (2015). Trissolcus japonicus (Ashmead) (Hymenoptera, Scelionidae) emerges in North America. Journal of Hymenoptera Research, 43, 119–128.
- Talamas, E. J., Johnson, N. F., & Buffington, M. (2015). Key to Nearctic species of Trissolcus Ashmead (Hymenoptera, Scelionidae), natural enemies of native and invasive stink bugs (Hemiptera, Pentatomidae). Journal of Hymenoptera Research, 43, 45–110.
Photo Credits
- Map courtesy of stopbmsb.org
- Image courtesy of Jeff Wildonger, USDA-ARS-BIIR;
- Images courtesy of Zach Schumm & Kate Richardson, Utah State University
- Image courtesy of Rutgers New Jersey Agricultural Experiment Station
- Image courtesy of Ashley Jones, University of Maryland
- Image courtesy of John Rosenfeld, bugguide.net
- Image courtesy of John Rosenfeld, bugguide.net
- Image courtesy of Kate Richardson, Utah State University
- Image courtesy of Elijah J. Talamas, USDA ARS
- Image courtesy of Kate Richardson, Utah State University
- Image courtesy of Cody Holthouse, Utah State University
- Image courtesy of Kate Richardson, Utah State University
- Images courtesy of Cody Holthouse & Zach Schumm, Utah State University
- Image courtesy of Kate Richardson, Utah State University
Funding
Funding for this publication was made possible by the National Institute of Food and Agriculture, U.S. Department of Agriculture, Specialty Crop Research Initiative (under award number 2016-51181-25409), U.S. Department of Agriculture, Utah Agricultural Experimental Station, Animal and Plant Health Inspection Service, Plant Protection and Quarantine (USDA-APHIS-PPQ) (under cooperative agreement number AP18PPQFO000C197), and USU Extension.
Related Research




















