Fact Sheet: Kratom

Common Myths and Facts About Kratom
There are many common myths and misunderstandings associated with kratom. Below are some of the commonly heard myths (U.S. Food and Drug Administration [FDA], n.d.; Heywood et al., 2024; Rogers et al., 2024; Suhaimi et al., 2025).
Myth 1. Kratom is not dangerous. False.
Kratom’s use is linked to cardiac arrest, liver toxicity, and other serious side effects. 7-OH, a naturally occurring opioid substance derived from the kratom plant. Also known as “legal morphine,” 7-OH has been described by the FDA as a threat to public health.
Myth 2. If kratom was unsafe, it would be illegal to sell in Utah. False.
The Kratom Consumer Protection Act in Utah passed in 2019 was designed to establish quality and purity standards. Residents over 21 can purchase products. The Act only requires the following.
- Registration: Retailers and manufacturers must register their establishments with the Utah Department of Agriculture and Food (UDAF) to sell kratom products.
- Product standards: All kratom products sold must meet specific safety and labeling standards to prevent contamination and ensure quality.
- Consumer safety: The act is designed to safeguard public health by ensuring that kratom products are unadulterated and accurately labeled.
Myth 3. If they sell it in a store, it must be FDA regulated. False.
Kratom is not regulated. According to the FDA (2024a), “… kratom is not lawfully marketed in the U.S. as a drug product, a dietary supplement, or a food additive in conventional food.”
Myth 4. Kratom will help with symptoms of withdrawal from any drug, especially heroin. False.
There is little evidence to support the use of kratom for these purposes.
Myth 5. It's fine to use; it is a “legal hallucinogen.” False.
Kratom is a hallucinogen with possible dangerous side effects. Kratom is an herbal substance derived from the leaves of a tropical tree in Southeast Asia, and it can produce both stimulant- and opioid-like effects. At higher doses, it can cause hallucinations and other psychotic symptoms, leading it to be sometimes referred to as a "legal hallucinogen."
Read on to Learn More
Below, we explore additional detail about what kratom is, discuss the ingredients in current products, adverse effects, and current regulations. Continue reading for additional information and key concerns.
What Is Kratom?
Kratom (Mitragyna speciosa) is a tropical tree native to Southeast Asia. For centuries, people in that region have chewed its leaves or brewed them into tea for the mild energy boost (stimulant) and opioid-like pain-relieving effects.
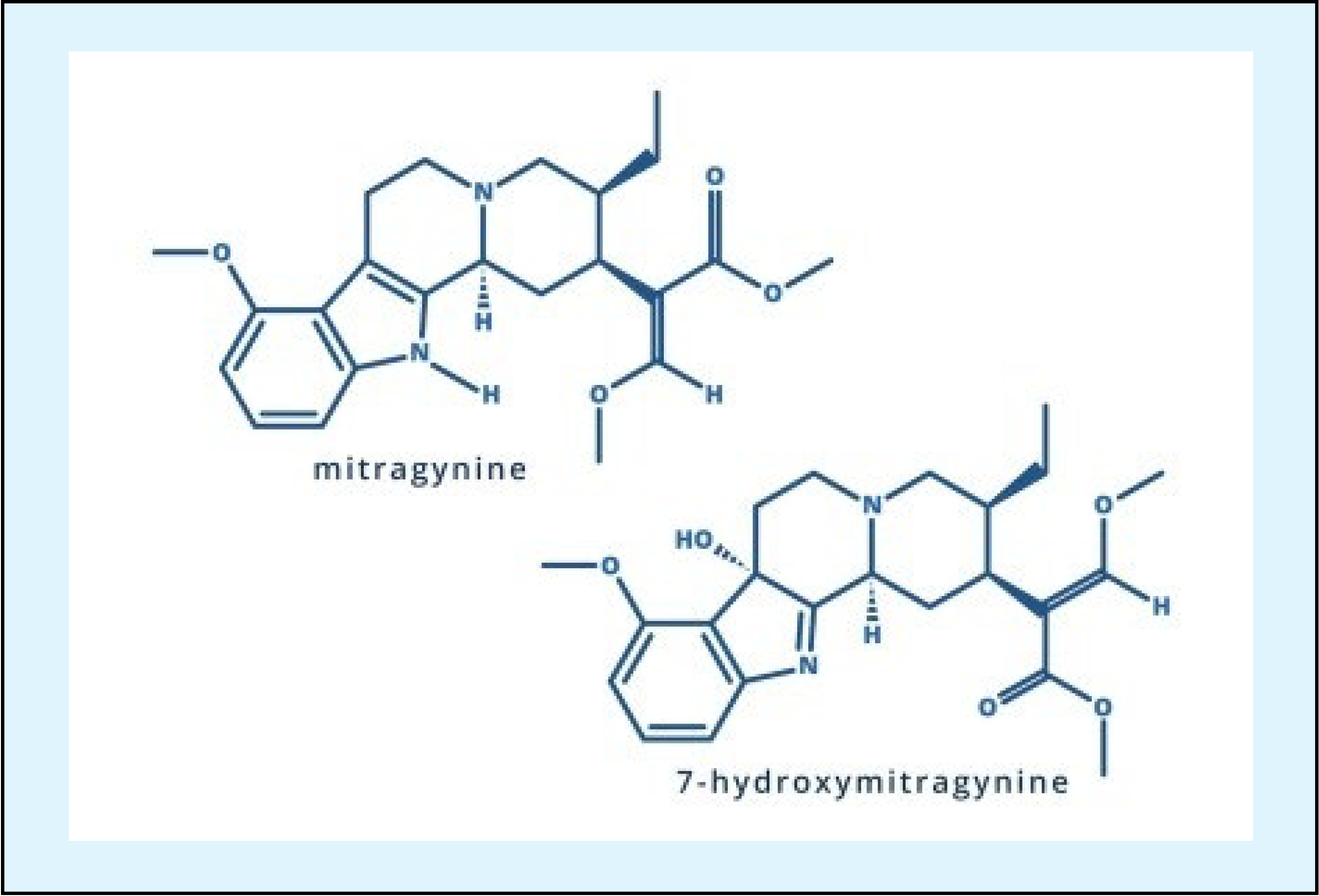
In recent years, kratom use has grown in the United States. It is sold in various forms, including capsules, powders, and most commonly as liquid extracts or “shots” (Heywood et al., 2024). These kratom products are promoted as health supplements, advertised for benefits such as boosting energy, enhancing mood, or producing opioid-like effects. However, most users may not fully understand what they are consuming and the potential risk they carry. Commercial kratom products vary widely in their chemical composition. Synthetic (human-made) or highly concentrated extracts may contain unpredictable levels of alkaloids (i.e., organic plant materials that have physical or psychological impacts on people) and have undergone little or no safety testing. See Figure 1 for active compounds (Hill et al., 2025) and Figure 2 for a 2D diagram of kratom’s chemical structure (National Institute on Drug Abuse [NIDA], 2022).
Figure 1. Active Compounds of Kratom
- Kratom can have opioid-like, analgesic, or stimulant effects depending on what dosage and compounds are used.
- The active compounds in kratom are called alkaloids. Mitragynine and 7-hydroxymitagynine (7-OH) are the most abundant.
- Mitragynine stays in the body for nearly 12 hours, and 7-hydroxymitragynine stays in the body for about 6 hours.
- Kratom infiltrates the blood stream and then flows to organs like the brain, liver, kidneys, and lungs. The compounds bind to the brain’s opioid receptors, similar to scheduled opioids, and are stronger than codeine but weaker than fentanyl and morphine (Heywood et al., 2024).
- Depending on how kratom is grown, it can have a different alkaloid content (Heywood et al., 2024). Synthetic 7-OH has opioid-like effects with limited safety testing that are not found in the leaves or extracts and are potential public health concerns (Hill et al., 2025; Smith et al., 2024).
- 7-OH far exceeds what is in the kratom leaves themselves, and there are no FDA-approved drugs with kratom or 7-OH for therapeutic use (Reissig et al., n.d.).
Credit: NIDA, 2022
Kratom is often used for self-management of pain, opioid withdrawal, and various other health conditions, with some individuals taking it as a recreational substance due to its stimulating and euphoric effects (Heywood et al., 2024; Suhaimi et al., 2025). However, there are many risks associated with its use (Figure 3), including concerns with cognitive performance, developing dependence, and potential for overdose (Suhaimi et al., 2025).
Figure 3. Product Variability of Kratom
- Kratom’s alkaloid content varies by growth and processing.
- Products sold across the U.S. often list “servings” that range from 1–15+ per bottle; consumers frequently drink the entire bottle, unintentionally taking many servings at once (Rogers et al., 2024; Heywood et al., 2024; McCurdy, 2025).
- If you do use these products, be sure to check the serving size to protect yourself.
- Kratom has altered the way patients have responded to opioids, resulting in some needing higher doses to get the same amount of pain relief (Akid et al., 2025).
- It can significantly impact the way prescription medications or illicit substances interact in the body; however, further research is required to decipher the exact mechanisms involved.
- Many kratom products have inadequate instructions for use or the amount of the active ingredients are in them, making it hard for users to know the product strength they are consuming (Heywood et al., 2024).

Supplement Facts
Serving Size: 10 mL
Servings Per Container: 3
Photo credit: ChatGPT
Kratom and 7-OH Shots: What to Know
7-hydroxymitragynine (7-OH) is emerging as a potent, engineered opioid-like derivative often sold as “kratom” or “natural extract.” It can be up to 13 times more potent than morphine and is now found in supplements sold in convenience stores, gas stations, vape shops, and online shops across the U.S. (FDA, 2025d)—places that never used to carry opioids. These products are marketed as harmless supplements but act like powerful opioids, creating serious risks for addiction, withdrawal, and overdose.
7-OH and kratom products are disguised as candy, gummies, or “energy shots.” Because they’re not prescription drugs, anyone—including teens—can buy them. This easy access makes 7-OH products a public health threat and a likely next wave in the opioid overdose crisis (FDA, 2025d).
About These Ingredients
Mitragynine and 7-hydroxymitragynine (7-OH) are the main active chemicals in kratom. They act on the brain’s opioid receptors, which can reduce pain but also cause dependence, sedation, and withdrawal if used often or in high doses (Heywood et al., 2024; Suhaimi et al., 2025). 7-OH is much stronger than mitragynine, so even small increases can greatly raise the risk of overdose or side effects (Hill et al., 2025). While kratom leaves naturally contain very small amounts of 7-OH, many “enhanced” or “spiked” products contain 5 to 10 times more than natural levels (FDA, 2025d). These concentrated forms are not the same as natural kratom and carry much higher risks of substance use disorder, withdrawal, and overdose. Figure 4 shows a sample supplement bottle and key factors.
The following example in Table 1 shows how kratom “shots” may appear safe but actually deliver opioid-level doses when consumed all at once.
Table 1. Ingredients in Kratom by Amount per Serving, Bottle, and What to Watch For
| Ingredient | Amount per serving (10 mL) | Total per bottle (30 mL) | Recommendation/What to watch for |
|---|---|---|---|
| Mitragynine | 75 mg | 225 mg | Keep under 100 mg per serving (no more than 300 mg total per day) to lower risk of nausea, dizziness, and dependence (Heywood et al., 2024). |
| 7-Hydroxymitragynine (7-OH) | 3 mg (4%) | 9 mg | The 7-OH amount should be under 2% of total alkaloids. Higher levels suggest a concentrated or synthetic product with greater overdose risk (Hill et al., 2025). Avoid products that do not list 7-OH content. |
| Caffeine | 60 mg | 180 mg | More than 200 mg per bottle can cause jitters, rapid heart rate, or anxiety—especially when combined with stimulants or if experiencing dehydration. |
| Alcohol | 10% | — | Avoid kratom mixed with alcohol; the combination slows breathing and increases overdose risk (Torrico et al., 2024). Choose products labeled “alcohol-free.” |
Quick Takeaways
- Always check how many servings are in the container and multiply each ingredient amount by that number to see what you will take of each ingredient.
- More is not better, and large doses increase the risk of nausea, dizziness, or dependence (Rogers et al., 2024).
- Avoid any kratom shot labeled “super concentrated,” “7-hydroxy,” or “legal high” and those with more than 2% of 7-OH, indicating elevated levels and higher risk.
- Mixing kratom with alcohol, pain medications, or sleep aids increases overdose risk (Torrico et al., 2024; FDA, 2024a).
- Even when labeled “natural,” kratom acts on opioid receptors and can cause dependence or overdose (Suhaimi et al., 2025).
- For questions or emergencies, call Utah Poison Control: 1-800-222-1222.
Adverse Effects
There are many potential adverse effects from using kratom. Some common effects follow.
- Possible toxicities: A possible increase in liver toxicity, seizures, substance use disorder, and death from a combination of kratom and other substances has been observed (FDA, 2024a; Heywood et al., 2024).
- Common effects among whole-leaf kratom products: These products may lead to or increase cravings, low energy, irritability, fatigue, anxiety, restlessness, difficulty sleeping, and negative mood symptoms (Rogers et al., 2024).
- Withdrawal: In most cases of kratom use, people can experience mild to moderate withdrawal symptoms on the Subjective Opiate Withdrawal Scale. Those who have experienced severe symptoms used kratom around six times per day. Males tended to experience more immediate effects after using kratom and reported more serious withdrawal symptoms. Withdrawal symptoms increase when amount and dosage increase, but a higher frequency of dosing makes withdrawal worse (Rogers et al., 2024).
- Overdose: Experiencing an overdose from kratom, while less likely than from an opioid, is possible (Heywood et al., 2024). Naloxone can temporarily reverse an opioid overdose and some kratom overdoses. When using naloxone to reverse a kratom overdose, oxygen levels may drop again after 12 to 24 hours, which makes it important to pay continued close attention to the person after naloxone has been administered (Ahmed et al., 2023). Sometimes, naloxone is ineffective for kratom, and the patient needs to receive lipid infusion, labetalol, or other treatments (Peran et al., 2023). It is difficult to screen for kratom, so doctors treating an overdose may be unaware of the substance use (Wright et al., 2021; Ahmed et al., 2023). Using kratom with opioids, benzodiazepines, or over-the-counter medicines (e.g., acetaminophen, caffeine) increases the risk of opioid overdose and death (Torrico et al., 2024).
- Pregnancy: Not much is known about what happens to people who are pregnant and use kratom. Of the cases that are known, babies have been born with neonatal abstinence syndrome (NAS; Wright et al., 2021). NAS occurs when an infant experiences withdrawal symptoms after birth. Babies can have a risk for premature birth, a lower birth weight than usual, and grow at a slower rate than children who were not exposed to substances in the womb (Anbalagan et al., 2024). Infants can also experience jitteriness, irritability, and muscle stiffness after birth (FDA, 2024). Severe conditions can lead to seizures and infant death (Anbalagan et al., 2024). Mothers who used kratom before pregnancy felt like they needed to use kratom to feel normal and stated that they experienced opioid-like withdrawal symptoms when trying to discontinue using (Wright et al., 2021).

kratom product sold in
stores or online.
Credit: FDA, 2024b
What Are the Regulations?
With substances sold over-the-counter, be aware of the regulations and safety limitations that contribute to kratom sales and use (see Figure 5 for a real-life example product image).
The following key regulations can help you better understand these factors.
- Utah law: In Utah, places that sell kratom must follow rules to ensure the kratom they provide is registered and in compliance with the law. Sellers must ensure their product doesn’t contain harmful ingredients or illegal substances. It cannot have any lab-made substances that the kratom plant does not have. Containers need labels listing the 7-hydroxymitragynine (7-OH) concentration, which must be less than 2% of the alkaloid content. This law is meant to protect Utah consumers from dangerous kratom (Utah Code § 4-45-104, 2019). This law may change in the future; check for updates following November 2025.
- U.S. Food and Drug Administration (FDA) statements: The FDA has not approved prescription or over-the-counter medications with the main chemical substances in kratom and kratom itself. No applications have been sent to the FDA to test or review kratom’s safety or effectiveness. Until then, the FDA is warning people not to use it for treating any conditions. There is inadequate data to guarantee kratom’s safety in food additives or supplements without causing dangerous side effects. Kratom is not safe as a dietary supplement, substance, or food additive (FDA, 2024a).
- Marketing: Kratom has been marketed to be a substitute for opioids and to help manage opioid withdrawal symptoms. There have also been unsubstantiated claims that it is not habit-forming and that there is no risk to using it, despite evidence of physical dependence among some who use the products (Smith et al., 2024; Wright et al., 2021). Several companies have received letters from the FDA because they use wording to describe their product as a substance for euphoric effects, even though it is unapproved for safety and effectiveness. Companies have faced consequences for marketing and labeling that could appeal to minors. Kratom has been marketed as a safe dietary supplement, but there is not enough data to support that claim (Reissig et al., n.d.; FDA, 2025a; FDA 2025b; FDA, 2025c).
Protect Yourself and Others
Avoid buying any products that list “7-OH,” “hydroxy,” or “enhanced kratom.” These are engineered to be opioid-like, not natural plant products. Read packaging carefully—some are disguised as candy or gummies. If you or someone else experiences confusion, slowed breathing, or loss of consciousness after taking a kratom or 7-OH product, call 911 or Utah Poison Control at 1-800-222-1222.
Conclusion
Kratom has grown in popularity, but the synthetic 7-OH products continue to have many adverse effects and are not FDA-regulated. The availability and ease of access make this substance particularly dangerous because many people may be unaware of these effects. Before taking any new supplements or medications, it is always best to consult with your medical provider, and this is true with kratom or any related products (e.g., 7-OH) as well.
Additional Resources
- What Is Kratom? (USU Extension HEART Initiative video)
- Kratom (National Institute on Drug Use (NIDA)
- The Concerning Emergence of “Gas Station Heroin”(Psychology Today)
- FDA and Kratom (FDA)
- Hiding in Plain Sight: 7-OH Products (FDA)
- Neonatal Abstinence Syndrome (NAS): What You Need to Know (USU Extension fact sheet)
- Pregnancy and Substance Use (USU Extension fact sheet)
References
Ahmed, S., Tran, Q. V., & McLean, M. (2023). The great imitator: A case of accidental kratom overdose. Cureus, 15(8), e43144. https://doi.org/10.7759/cureus.43144
Akid, I., Roach, C., & Allan, T. (2025). The kratom bomb. Journal of Pain and Symptom Management, 69(5), e566. https://doi.org/10.1016/j.jpainsymman.2025.02.224
American Kratom Association. (2022). Consumer guidelines for kratom use. Retrieved November 10, 2025, from https://www.americankratom.org/news/consumer-guidelines-for-kratom-use
Anbalagan, S., Falkowitz, D. M., & Mendez, M. D. (2024, April 1). Neonatal abstinence syndrome. In StatPearls Publishing. National Library of Medicine. https://www.ncbi.nlm.nih.gov/books/NBK551498/
Gold, M. S. (2025, June 3). The concerning emergence of “gas station heroin.” Psychology Today.
Heywood, J., Smallets, S., & Paustenbach, D. (2024). Beneficial and adverse health effects of kratom (Mitragyna speciosa): A critical review of the literature. Food and Chemical Toxicology, 192, 114913. https://doi.org/10.1016/j.fct.2024.114913
Hill, K., Boyer, E. W., Grundmann, O., & Smith, K. E. (2025). De facto opioids: Characterization of novel 7-hydroxymitragynine and mitragynine pseudoindoxyl product marketing. Drug and Alcohol Dependence, 272, 112701. https://www.sciencedirect.com/science/article/abs/pii/S0376871625001541
Huberman, A. (Host). (2025, July 21). Health effects & risks of kratom, opioids & other natural occurring medicines, Dr. Chris McCurdy [Audio podcast]. Huberman Lab. https://www.hubermanlab.com/episode/health-effects-and-risks-of-kratom-opioids-and-other-natural-occurring-medicines-dr-chris-mccurdy
National Institute on Drug Abuse [NIDA]. (2022, March 25). Kratom. Retrieved October 16, 2025, from https://nida.nih.gov/research-topics/kratom#kratom-compounds
Peran, D., Stern, M., Cernohorsky, P., Sykora, R., Popela, S., & Duska, F. (2023). Mitragyna speciosa (kratom) poisoning: Findings from ten cases. Toxicon, 225, 107054. https://doi.org/10.1016/j.toxicon.2023.107054
Reissig, C. J., Chiapperino, D., Seitz, A., Lee, R., Radin, R., & McAninch, J. (n.d.) 7-Hydroxymitragynine (7-OH): An assessment of the scientific data and toxicological concerns around an emerging opioid threat. U.S. Food and Drug Administration (FDA). https://www.fda.gov/media/187899/download?attachment
Rogers, J. M., Weiss, S. T., Epstein, D. H., Grundmann, O., Hill, K., & Smith, K. E. (2024). Kratom addiction per DSM-5 SUD criteria, and kratom physical dependence: Insights from dosing amount versus frequency. Drug and Alcohol Dependence, 260, 111329. https://doi.org/10.1016/j.drugalcdep.2024.111329
Smith, K. E., Boyer, E. W., Grundmann, O., McCurdy, C. R., & Sharma, A. (2025). The rise of novel, semi‐synthetic 7‐hydroxymitragynine products. Addiction, 120(2), 387–388. https://onlinelibrary.wiley.com/doi/full/10.1111/add.16728
Suhaimi, F. W., Mohamad Khari, N. H., Hassan, Z., & Müller, C. P. (2025). Exploring the cognitive effects of kratom: A review. Behavioural Brain Research, 480, 115387. https://doi.org/10.1016/j.bbr.2024.115387
Torrico, T., Patel, K., Nikolov, N., Salam, M. T., Padhy, R., & Weinstein, D. (2024). Presence of kratom in opioid overdose deaths: Findings from coroner postmortem toxicological report. Frontiers in Psychiatry, 14, 1332999. https://doi.org/10.3389/fpsyt.2023.1332999
U.S. Food and Drug Administration (FDA). (n.d.). Hiding in plain sight: 7-OH products. https://www.fda.gov/news-events/public-health-focus/hiding-plain-sight-7-oh-products
U.S. Food and Drug Administration (FDA). (2024a, August 5). FDA and kratom. U.S. Department of Health and Human Services. https://www.fda.gov/news-events/public-health-focus/fda-and-kratom
U.S. Food and Drug Administration (FDA). (2024b, July 26). FDA warns consumers not to use optimized plant mediated solutions (OPMS) black liquid kratom. https://www.fda.gov/food/alerts-advisories-safety-information/fda-warns-consumers-not-use-optimized-plant-mediated-solutions-opms-black-liquid-kratom
U.S. Food and Drug Administration (FDA). (2025a, June 25). Warning letter to 7Tabz Retail, LLC (MARCS‑CMS 709546) [Warning letter]. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/7tabz-retail-llc-709546-06252025
U.S. Food and Drug Administration (FDA). (2025b, June 25). Warning letter to Hydroxie, LLC (MARCS-CMS 709661) [Warning letter]. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/hydroxie-llc-709661-06252025
U.S. Food and Drug Administration (FDA). (2025c, June 25). Warning letter to Royal Diamond Imports, Inc. (MARCS-CMS 709540) [Warning letter]. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/royal-diamond-imports-inc-709540-06252025
U.S. Food and Drug Administration (FDA). (2025d, March). Preventing the next wave of the opioid epidemic: What you need to know about 7-OH. U.S. Department of Health and Human Services. https://www.fda.gov/drugs/information-consumers-and-patients-drugs/hiding-plain-sight-7-oh-products
Utah Code § 4‑45‑104 (2019). Kratom processor requirements—Criminal penalty. In Chapter 329, General Session, Utah Legislature. https://le.utah.gov/xcode/Title4/Chapter45/4-45-S104.html#:~:text=Kratom%20processor%20requirements%20%2D%2D%20Criminal%20penalty.,-(1)&text=(4)-,A%20kratom%20processor%20may%20not%20prepare%2C%20distribute%2C%20sell%2C%20or,in%20accordance%20with%20this%20chapter.&text=A%20kratom%20processor%20shall%20register,Section%204%2D5%2D301
Utah State University Extension (2025, November 5). What is kratom? [Video]. YouTube.
Withers, E., Thompson, K., Hamilton, E., & Wright Voss, M. (n.d.). Pregnancy and substance use [Fact sheet]. Utah State University Extension. https://extension.usu.edu/heart/research/pregnancy-and-substance-use
Wright Voss, M., Van Komen, A., Hamilton, E., Cleveland, A., & Miller, J. (2023, May). Neonatal abstinence syndrome (NAS): What you need to know [Fact sheet]. Utah State University Extension. https://digitalcommons.usu.edu/extension_curall/2342
Wright, M. E., Ginsberg, C., Parkison, A. M., Dubose, M., Sherbondy, M., & Shores, E. (2021). Outcomes of mothers and newborns to prenatal exposure to kratom: A systematic review. Journal of Perinatology: Official Journal of the California Perinatal Association, 41(6), 1236–1243. https://doi.org/10.1038/s41372-021-00952-8
The authors of this content used ChatGPT to create the Figure 4 image. Authors reviewed and edited the image provided by the AI tool, and they take full responsibility for the content.

Utah State University Extension
Peer-reviewed fact sheet
Authors
Cadence Johnnie, Ashley Yaugher, Aaron Hunt, Cara Murray, and Tim Keady




